The Zero-order Rate Law calculator computes the rate of a reaction not dependent on the concentration of its reactants.
INSTRUCTIONS: Enter the following:
- ([A]) Concentration of A
- (k) Constant in (mol/L*sec)
Rate: The calculator returns the rate in moles per liter per second.
Chemistry Rate Law Calculators
- Zero Order Rate Law (Integral form)
- Zero Order Half Life
- Zero Order Rate Law
- First Order Rate Law (Integral form)
- First Order Half Life
- First Order Rate Law
- Second Order Rate Law (Integral form)
- Second Order Half Life
- Second Order Rate Law
The Math / Science
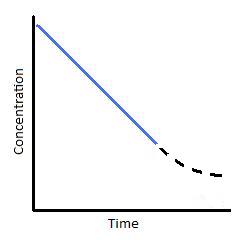
The Zero-order rate law[1] equation calculates the rate of a reaction that does not depend on the concentration of its reactants. Since the rate is independent of the reactant's concentration the rate will be constant but only of a certain amount of time. This rate reaction observation is confirmed by the theory of Michaelis-Menten kinetics[3], where a catalyzed reaction will have a constant rate up until a certain point in time where the rate starts to decrease / This is due to less substrate (reactants) binding to the catalyst.
The Math
The equation is:
Rate = k[A]0 = k
Where
- [A] is the concentration of substance A in units (mol/L)
- k is the rate constant in units of (mol/L*s)
Related Topics
Supplement Material
- Khan Academy: Rate law and reaction order
References
[1]https://en.wikipedia.org/wiki/Rate_equation
[2]Whitten, et al. 10th Edition. Pp. 626,629,631
[3]https://en.wikipedia.org/wiki/Michaelis–Menten_kinetics
[Graph]http://chemwiki.ucdavis.edu/Core/Physical_Chemistry/Kinetics/Reaction_Rates/Zero-Order_Reactions
